
29th of May 2019
Depression and Parkinson’s
A common duo in the spotlight
It is extremely important that neurologists realise that depression and cognitive problems are a big thing in Parkinson’s. Many neurologists are still insufficiently aware of this. In fact, some neurologists say: “This kind of thing is not for me”. But it really belongs to Parkinson’s | Monique Timmer
Parkinson’s disease is characterized by a massive loss of dopamine-producing neurons in a small core in the brain stem, the substantia nigra (2).
Dopamine is a neurotransmitter, a signal substance that transmits impulses from nerve cell to nerve cell. Our brain controls our movements with these signals. Movements that range from walking, talking, laughing and writing to, for example, the contraction of stomach and intestinal muscles. As the degeneration of the substantia nigra progresses, it becomes increasingly difficult to control movements. This leads to all sorts of motor symptoms, such as slowness of movement, stiffness, and tremor.

That is not the whole story. Neurodegeneration in patients with Parkinson’s disease is not limited to the substantia nigra. Other areas of the brain are also affected to a greater or lesser extent and this impairment, in addition to the damage to the dopaminergic system, contributes to the development of so-called non-motor symptoms, such as sleep problems and many neuropsychological symptoms such as problems in thinking, anxiety disorders, apathy, and depression. Depression is one of the most common non-motor symptoms in Parkinson’s.
In patients with depression, for example, the loecus coerulus and raphne nuclei have been demonstrably affected. Noradrenaline-producing neurons are located in the locus coerulus and in the raphe nuclei serotonin-producing neurons reside.

Depression in patients with Parkinson’s disease is common. Estimates vary, but on average 35% of patients suffer from a clinically relevant depression. Yet depression is often not recognized as such. On the one hand, this is because part of the symptoms also occurs in Parkinson’s patients without depression. Consider, for example, the flattening of the facial expression, slow movement, softer talking, sleep disturbances, a lack of energy and a reduced ability to pay attention to something. If we add to this that patients themselves do not so easily admit that they experience depressive symptoms, then it is clear that depression is often overlooked.
To diagnose depression in people with Parkinson’s disease, it is important to look behind the mask of external symptoms.

Dopaminergic means “related to dopamine”. Dopaminergic medication increases dopamine-related activity in the brain. The most commonly prescribed dopaminergic drugs are levodopa, a dopamine precursor substance, and the so-called dopamine agonists. These substances fit on the dopamine receptor, are able to stimulate it and thus mimic the action of dopamine. Well-known dopamine agonists are ropinirole, pramipexole, rotigotine, and apomorphine.

Part of the cause of depressive symptoms lies in changes in the dopaminergic system. We know that because some patients already experience depression or burnout before they are diagnosed with Parkinson’s disease. Their mood often improves as soon as they start taking dopaminergic medication. In addition, 75% of patients not only experience response fluctuations of the medication to their motor symptoms but they also experience so-called mood swings (non-motor fluctuations). If the medication runs out, and patients are OFF, not only do their motor symptoms worsen but also their depressive symptoms.
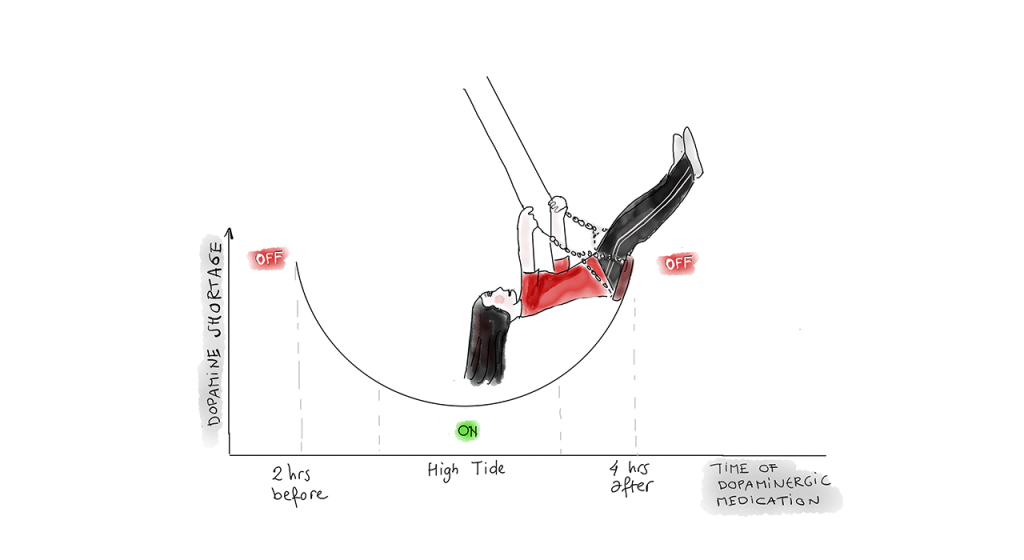
However, not all depressions are the same. The role of the dopaminergic system is greater in some patients than in others. We do not yet know why that is the case. In addition, other neurotransmitter systems, such as the noradrenergic and serotonergic systems, also play an important role in depression. What exactly this role is and how the different neurotransmitter systems influence each other in depression in Parkinson’s disease has not yet been clarified.
In daily life, we adjust our behavior based on our experiences. Patients with Parkinson’s disease and previous or current depression appear to be less good at learning from positive experiences, while they are just as good at learning from negative experiences as patients without depressive symptoms.
I will give an example: Suppose you have bad experiences with a Sushi restaurant. If a colleague proposes to go to that restaurant, you still go and to your great surprise, the food is now very tasty. You receive an unexpected reward and adjust your association. If you decide to go again next time, you go with high expectations. This time the food turns out to be very distasteful again. It turned out that the last time a substitute cook had been working and he has now left the restaurant again. You get an unexpected negative experience and adjust your association again.
Parkinson’s patients with depressive symptoms are worse in reversing the association from a negative association to a positive association. If we look at the example of the Sushi restaurant again, this could mean, for example, that the patient in question does not adjust his association with the Sushi restaurant when the food turns out to be tastier than expected. The next time he or she would still skip the restaurant.
This is just an example to illustrate the principle. The magnitude of the effect varies per patient. My research results only indicate that there is a relationship between depression and a reduced ability to learn from a reward. We also see this behavior in people with depression without Parkinson’s by the way.

Dopaminergic medication can improve or deteriorate cognitive functions. The moment you receive dopaminergic medication, the amount of dopamine in a brain area called the dorsal striatum replenishes. That makes it easier to move again. In addition, the cognitive functions associated with this brain area improve, such as being able to shift your attention. But the medication also ends up in other areas of the brain. If the dopamine ends up in areas of the brain that do not have a dopamine deficiency, then this can lead to an overdose, with the result that certain cognitive functions deteriorate.
In my research, I looked at the ability of patients to reverse their associations with the CANTAB-IED test. What was ‘right’ at first changed to ‘wrong’ and vice versa. This cognitive ability is associated with a brain region called the ventral striatum. In patients with depression, this area is thought to have a dopamine deficiency. Patients without depression do not exhibit this deficiency.

I observed that patients who took more dopaminergic medication were worse at reversing these types of associations. This is in line with the overdose theory, in which the ventral striatum overflows with dopamine, resulting in various negative cognitive effects. However, Parkinson’s patients with depression turned out to score the worst. We did not expect this result since the brain area in question in these patients is associated with a dopamine deficiency. Apparently, other mechanisms also play a role, such as, for example, being able to keep the dopamine level properly in balance and also the balance between dopamine and other neurotransmitter systems, such as the serotonergic system, has its role to play.
But even though the underlying mechanisms are still unknown, it is still an important discovery for the neurologists’ treatment room. Based on these outcomes, neurologists should be extra alert to the cognitive side effects of dopaminergic medication, especially in Parkinson’s patients with depression.
We subjected patients to a gambling game, specially designed to measure their aversion to loss. The participants could win between 6 euros and 30 euros and lose between 3 and 15 euros at every bet, head or coin, as it were. This set-up is based on the assumption that people are on average twice as sensitive to loss than to profit. We tested patients with and without depression twice. Once when they were ON, after taking medication, and once after not taking medication for at least 18 hours.
Patients without depression and without medication gambled less. The amount of money they were gambling for didn’t influence this decision. Patients with depression also gambled less without medication, but within the bets they placed they were relatively more sensitive to loss. The profit had to be relatively higher for them to take on the bet. Patients with and without depression and without medication therefore both appear to be more risk-averse, but through different mechanisms. I say appear because we could not formally compare this with healthy test subjects.

After taking dopaminergic medication, the behavior of both groups of Parkinson’s patients normalised. The medication, therefore, changes the decision of Parkinson’s patients to take a gamble or not. Parkinson’s patients without depressive symptoms more often accepted a gamble, regardless of the amount of money they were gambling for, while depressed patients more often accepted a gamble because the medication made them less loss averse.
We did a so-called Stroop test with our test subjects. For example, the test subjects would see an arrow pointing to the right with the word “LEFT” in it and vice versa. One time they were asked to respond to the arrow, the other time to the word in the arrow. Half of the tests were preceded by a 10-cent reward and the other half by a 1-cent reward.
Parkinson’s patients were able to switch their attention more easily from the direction of the arrow to the word in the arrow and vice versa if the reward was higher. This was the case in both patients with and without depression. This is in contrast to healthy test subjects, who made more mistakes with a higher reward. All in all, we can conclude that patients with Parkinson’s seem to benefit from a reward as a motivator to stimulate switching their attention to a different task.

Perhaps you can offer patients a reward. For example: “Let’s go for a walk to the park and grab an ice cream.” But in the end every patient and partner – and therefore every relationship – is different and everyone must find his or her own way. But it helps enormously if you can occasionally zoom out together to see what happens. And then to try out what works and what doesn’t.

It is extremely important that neurologists realise that depression and cognitive problems are a big thing in Parkinson’s. Many neurologists are still insufficiently aware of this. In fact, some neurologists say: “This kind of thing is not for me”. But it really belongs to Parkinson’s.
With my dissertation, I have demonstrated that we must be vigilant for the negative effects of dopaminergic medication on cognition, especially in patients with depression.The sooner you notice these types of effects, the sooner you can adjust.
Fortunately, more and more research is being done, but there is still a way to go. In the meantime, I contribute to raising awareness with presentations in the region, poster presentations at conferences and the scientific article: “What a neurologist should know about depression in Parkinson’s disease” (3).
Why is Monique’s work important to me?
The research by Monique Timmer offers wonderful starting points for a good discussion about the impact of neuropsychological symptoms in Parkinson’s disease. Making visible what is going on, can help you deal with it or even make you find a way around it.
The landscape of detours in motor freezing is already being documented. There are also general recommendations for maintaining cognitive health in Parkinson’s disease (4), but I have not yet seen a concrete overview of strategies and detours for various types of mental freezing. Mental freezing is more diffuse, less concrete, more of a taboo, less researched, less visible and also simply less sexy than motor freezing. That is unjustified because the impact of mental freezing on the quality of life is huge. After all, you can disappear as a person.
I have made it my personal mission to collect detours to cope with the neuropsychological symptoms of Parkinson’s. I hope to turn the insights I gain from such a collection into concrete tools which will increase understanding between patients and their loved ones. Sometimes, it’s very lonely in Parkinson’s country and it is really nice – or essential even – that a number of people can travel along to paint the new landscape together.
To get started with the grand “Mental freezing detours collection”, I am throwing in three detours from my own experience:
- Make your questions specific
Do not ask “How are you?” I really have no idea where to start.
Rather ask for something specific such as, “How is your son’s study going?” - Visualise all steps
In the middle of the forest – where there doesn’t seem to be any coffee bar in miles – do not propose to drink coffee together because I may not respond. My lack of response may seem unpleasant, but it is certainly not intended that way. In fact, I am looking very hard for a start. But there are so many roads in my brain that can lead to coffee that I would not know where to start.
What you cán do if you want a response is to spell out the steps. For example: ‘Shall we walk to the car, drive to your house and then drink a cup of coffee?’ You bet that I’ll give you an answer.
Sometimes the words don’t want to come to me. Then I move my hand from left to right over my forehead a number of times as a sign that I am scanning but receiving nothing. At home, we call the gesture ‘The Knight Rider‘. Humor gives air and helps to acknowledge and accept things as they are now. Another time will come.
Your turn
I think we could learn a lot from each other’s detours. That’s why I want to ask you a question:
Do you recognize that you sometimes get stuck in your thinking and feeling? That you simply cannot start with a cognitive task? And have you and your loved ones found detours around it? Let me know! If you scroll all the way down on this page, you can leave your tips in the comments. Or email me at sparks@verbeeldingskr8.nl. I am really curious!
With your input, I hope to be able to compile a nice list of mental freezing detours. Illustrated of course. That’s a promise : -)

Sparks
Sources
(1) Timmer, M.H.M. (2019). Out of Balance. Neurocognitive mechanisms underlying depression in Parkinson’s disease. [THESIS]. Retrieved from https://books.ipskampprinting.nl/thesis/529302-Timmer/ (Open Access)
(2) Parent, M., & Parent, A. (2010). Substantia Nigra and Parkinson’s Disease: A Brief History of Their Long and Intimate Relationship. Canadian Journal of Neurological Sciences / Journal Canadien Des Sciences Neurologiques, 37(3), 313-319. https://doi.org/10.1017/S0317167100010209. Retrieved from http://www.biusante.parisdescartes.fr/chn/docpdf/parent_10_2017%20(3).pdf (Green Open Access)
(3) , et al (2017). What a neurologist should know about depression in Parkinson’s disease. (Closed Access, or see Chapter 2 of Monique’s Thesis at https://books.ipskampprinting.nl/thesis/529302-Timmer/ for the Open Access version)
(4) Goldman, J.G., et al (2018). Cognitive impairment in Parkinson’s disease: a report from a multidisciplinary symposium on unmet needs and future directions to maintain cognitive health. npj Parkinson’s Disease 4, Article number: 19. Retrieved from https://doi.org/10.1038/s41531-018-0055-3 (Open Access)
About Monique Timmer

Monique Timmer (1982) studied medicine at the University of Utrecht. In 2008 she started her neurology training at the Radboud UMC in Nijmegen. In 2011 she started a PhD research on the neurocognitive aspects of Parkinson’s disease. Since October 2018 she has been working as a neurologist at the Radboud UMC and the Parkinson Center in Nijmegen.