Parkyvist* in the picture
Building upon Kevin McFarthing’s Hope list
10th of August 2020
When I was born into Parkinson’s it wasn’t long until I ran into an impressive ‘list of hope’ – bit.ly/ParkinsonsHopeList – gathered by Kevin McFarthing. Kevin’s Hope List has two main sections: research projects that are in the discovery and preclinical stages, and projects in the various phases of clinical trials. All with a focus on Parkinson’s disease.
The clinical trial section includes entries from Clinicaltrials.gov, manually gathered company planned trials, those already completed but waiting for the next steps to be announced, early trials yet to be registered, trials registered on other clinical trial registries, and some in the regulatory phase. And – importantly – the list of hope is free to be built upon!
Why did Kevin create the Hope List and what is he hoping for himself, 8 years after his diagnosis with Parkinson’s Disease? You can read all about it in the Q&A below. Below the interview, I try to make some of the hope speak to even more of my readers with some drawings and vizzes. This lengthy blog post will conclude with a plea on the importance of opening up data FAIRly (Three blogs for the price of one ;)).
Part 1. Where hope is born. A Q&A with Kevin
“If there isn’t a cure then where can I find one?” | Kevin McFarthing on why he started compiling his ‘Hope list’

I was born and raised in the North East of England. I’m married with two children and two grandchildren. I have a PhD in Biochemistry from the University of Liverpool. After University I worked for a company making products for use in life sciences research. These products could best be described as molecular tools. I then became Head of R&D for the diagnostics division of a large pharma company, developing tests for a variety of biomarkers.
The next step in my career was at Reckitt Benckiser as Head of R&D for Health & Personal Care. RB is a Financial Times Stock Exchange 100 (FTSE) top 20, multi-national consumer goods company with a large business in consumer medicines. At the time RB also had a sizeable prescription-only product for the treatment of opioid use dependence.
I then became Global Head of Strategic Alliances for the whole business, working with existing partners and bringing new ones on board, especially at the “front end” of innovation. When I left RB, I set up a consultancy business, Innovation Fixer Ltd, helping clients improve the effectiveness and efficiency of innovation management.
So – a career in industrial science with a healthcare focus.
I was diagnosed at 4pm on Monday 10th December 2012, a date I understandably remember well. I was 55 with no future view other than a long and healthy old age. The diagnosis was blunt, as was the accompanying advice – there’s no cure and come back in six months.
I’d like to think that I haven’t changed much since the diagnosis, but my family thinks I’m quieter than I used to be; I’m probably more reflective.
As soon as I was diagnosed I started to find out as much as I could about Parkinson’s. This included looking for therapies in the pipeline. It’s probably the same with a lot of newly-diagnosed PwP, “If there isn’t a cure then where can I find one?”
My research moved from a disorganised list in Evernote to an Excel spreadsheet which, to cut the story short, became the Parkinson’s Hope List. I shared the list with a few people who gave me positive feedback. I then started to circulate it more widely and now it’s freely accessible at bit.ly/ParkinsonsHopeList. There are now over 300 projects on the list all the way from discovery to regulatory submission.
Once the Hope List had been built, it has been relatively straightforward to keep it maintained. I see a lot of information from a lot of sources, particularly Simon Stott’s excellent www.scienceofparkinsons.com, (on Twitter at @ScienceofPD), but also Parkinson’s News Today. Twitter, of course, is a great source of Parkinson’s news, with one account to highlight, @Ingentium_PD.
I release updates of the Hope List around every three months, and I give it a thorough “clean” about twice a year. This part takes quite a bit of time, trying to find out whether companies still exist and whether Parkinson’s is still a priority for them. Since I started releasing the list in 2017, over 130 projects have dropped off the list, of which only seven came off because they were launched or submitted for regulatory review.
When I look at the list, I’m really impressed by the sheer numbers of projects – well over 300 – and the associated investment. The big gap is the lack of disease-modifying trials in the late stage, phase 3.
Disease-modifying trials (DMT) fail for several reasons: There is a lack of definable, biomarker-supported sub-types, which of course are an important reason why trials fail. But I think there are some more reasons. The UPDRS is unable to discriminate sufficiently well in longitudinal studies where there is an element of training and learning by patients. The placebo response can be very high. The placebo arm often doesn’t deteriorate very much, leaving too small a gap between placebo and active. The patient cohorts are either too wide (age 18-80, have PD) or too narrow (<60, drug-naive, need to stay off drugs). There are no validated biomarkers. It’s also not clear why all the spectacular phase 3 failures usually have a successful phase 2 trials behind them.
I’d like to see less dependence on the UPDRS as the primary outcome measure.
My symptoms have progressed relatively slowly, so I’d be very happy if something is found that stops that progression and, of course, the sooner the better. That would let me get on with the rest of my life focusing on life itself, not what gets in the way or might do so in the future.
I’ve engaged in a lot of patient-researcher collaboration and there are some superb scientists and clinicians engaging with informed, engaged and enthusiastic patients. Everybody benefits, from improved clinical trial protocols to the basic researcher who can say, when meeting a patient – “so that’s why my work is important”.
My vision for such collaboration is that both patients and researchers learn enough of the other’s language and experience to understand and empathise; to influence where it makes sense and to keep away when it doesn’t.
My advice for the newly-diagnosed has three Es:
- Educate yourself about the condition and what you can do about it yourself.
- Engage with Parkinson’s – it’s not going away any time soon so denial wastes valuable time.
- Exercise – this is the only therapy so far proven to slow the progression. Have a mix of aerobic exercise that gets you hot and sweaty together with movement exercise that keeps you supple (always consult your doctor first).
I’d also advise the newly-diagnosed to be very careful about a lot of self-claimed “miracle workers” who claim to be able to reverse or cure Parkinson’s; or who propose magic supplements with no proof of safety or efficacy. They’ll not only take a lot of your money, but potentially damage your health. Again, always consult your doctor first.
Part 2. Hope, filtered, drawn and visualised
Kevin and co-authors have just published an article about a selection of hope: The ‘drug pipeline’ ((McFarthing et al, 2020). They analyse the 145 drug trials from Clinicaltrials.gov which are testing ‘drugs’ (or ‘pharmacological treatments’ in jargon) in order to “raise awareness of the clinical trial landscape in PD, both with researchers and the wider PD community”. Kevin has written a reflection on his Medium space.
My vision for patient-researcher collaboration is that both patients and researchers learn enough of the other’s language and experience to understand and empathise | Kevin McFarthing in this blogpost.
For patients and researchers to truly learn each other’s language – as Kevin envisions – lots of bridges still have to be built. Bridges that not only raise awareness of the drug pipeline out there but also truly enable both patients and researchers to feel confident enough to meet and interact halfway.
To begin crossing the bridge towards the expertise Kevin and his co-authors have myself, I have drawn the 14 ‘drug pipeline categories’ which Kevin and his co-authors discern. What are the agents under investigation? What is their intended mode of action? I hope that my drawings, short explanations, and visualisations that follow will also help some of you find the common language needed to confidently walk up to the authors and interact from your unique perspective.
Hope drawn in 14 drug categories

I. MECHANISM OF ACTION: Antioxidant (n=2)
In healthy cells, there is a balance between the production of highly reactive free radicals including reactive oxygens species (ROS) which may damage cells and the level of antioxidant which break ROS down. In the case of oxidative stress, ROS takes control and overpowers the antioxidant army. Oxidative stress is implicated in the pathogenesis of many diseases. Parkinson’s disease is one of them. Antioxidants which are being tested in PD clinical trials are Hydrogen and Deferiprone (an iron-chelator).

II. AGENTS: Botanical (n=4)

III. MECHANISM OF ACTION: Cell therapy (n=9)

IV. MECHANISM OF ACTION: Dopaminergic symptom relief (n=34)
Trials in this category focus on either restoring, replacing, or mimicking the neurotransmitter dopamine. Examples of the focus of trials in this category are e.g. variation in the delivery dose and testing substances that prolong the effect of medication (e.g. subcutaneous administration of levodopa and carbidopa).
The authors also categorise a trial investigating AADC gene therapy under this category. The AADC enzyme – which is coded for by the AADC gene – is responsible for the final step in the synthesis of some key neurotransmitters such as dopamine. Gene therapy in this context aims to restore the ability to make dopamine in the striatum of the brain, rather than receive it from the substance nigra.

V. AGENTS: Energy and mitochondria (n=4)
Impairment of energy management in mitochondria is thought to play a role in the development of Parkinson’s disease. Trials in this category aim to investigate and ultimately restore mitochondrial function. Examples include the administration of gold nanocrystals to influence the NAD metabolism in mitochondria and the administration of ursodeoxycholic acid (UDCA) to increase the ATP levels in the brain. The ratio of NAD+ to NADH – two forms of nicotinamide adenine dinucleotide – dictates how effectively the cell can produce ATP. ATP – adenosine triphosphate – is the transporter of chemical energy within cells.

VI. MECHANISM OF ACTION: GBA (n=4)
This category is applied to agents that enhance the activity of glucocerebrosidase (GCase). GCase is an enzyme that helps digest the waste in the lysosomes of our cells. Mutations in GBA1, which codes for GCase, are a risk factor for the development of Parkinson’s. Genetic variations outside of the GBA gene can also influence GCase protein levels (Schierding, 2020).
In clinical trials, compounds with exotic names such as GZ/SAR402671 are tested in people with Parkinson’s with a known GBA mutation. Ambroxol is being trialed in two places pursuing two different purposes: disease modification and as a treatment for Parkinson’s disease dementia.
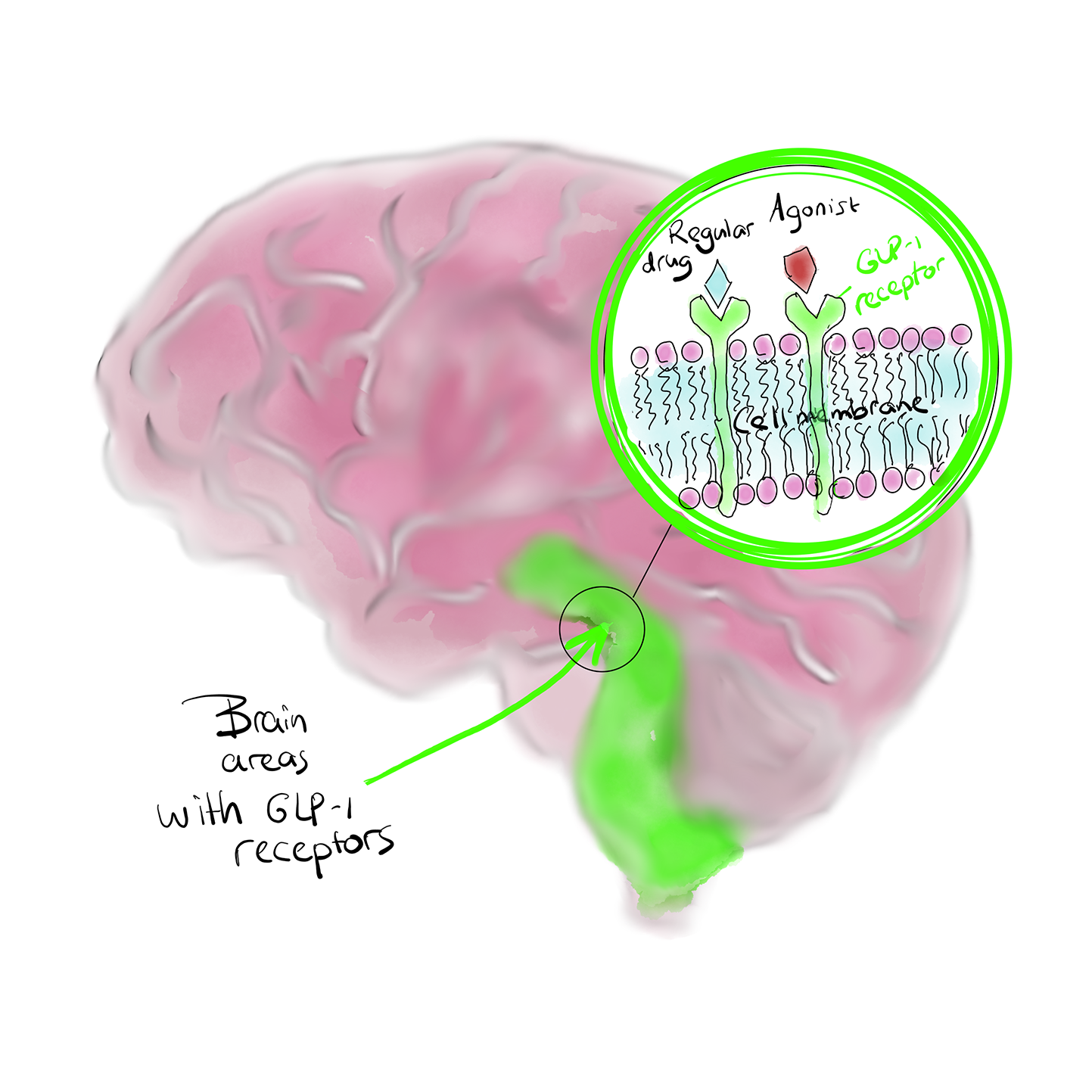
VII. MECHANISM OF ACTION: GLP-1 agonist (n=6)
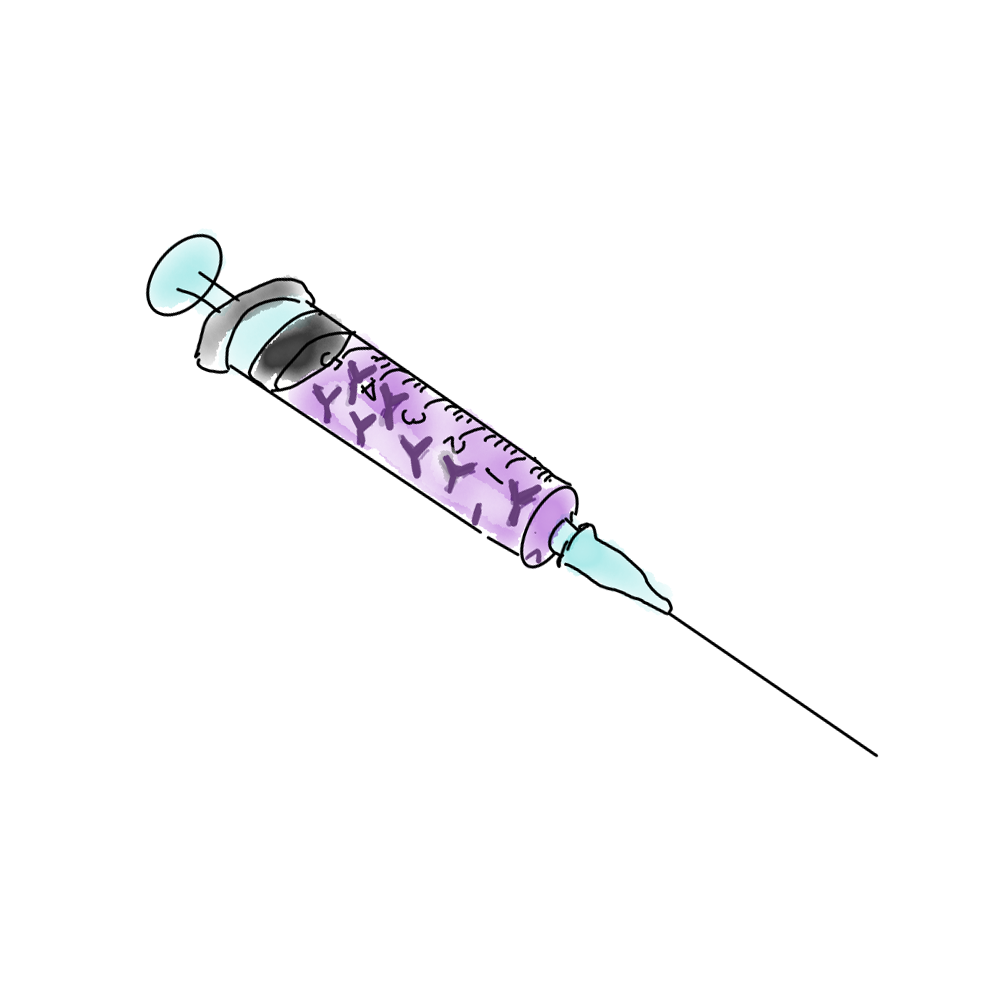
VIII. MECHANISM OF ACTION: Immunotherapy (n=7)

IX. MECHANISM OF ACTION: Kinase inhibitor (n=4)
Protein kinases catalyse protein phosphorylation, one of the major mechanisms that regulates cellular activities. (Mehdi, 2016). Kinase inhibitors represent a broad category encompassing agents blocking specific kinase activity. E.g. mutations in the LRRK2 gene – coding for Leucine-Rich Repeat Kinase – lead to overexpression which is implicated in the pathogenesis of Parkinson’s Disease. Some examples: An LRRK2 inhibitor is being tested in a clinical trial. Another kinase inhibitor is inhibiting c-Abl tyrosine kinase. c-Abl tyrosine kinase activation leads to oxidative stress and is associated with many neurodegenerative diseases.
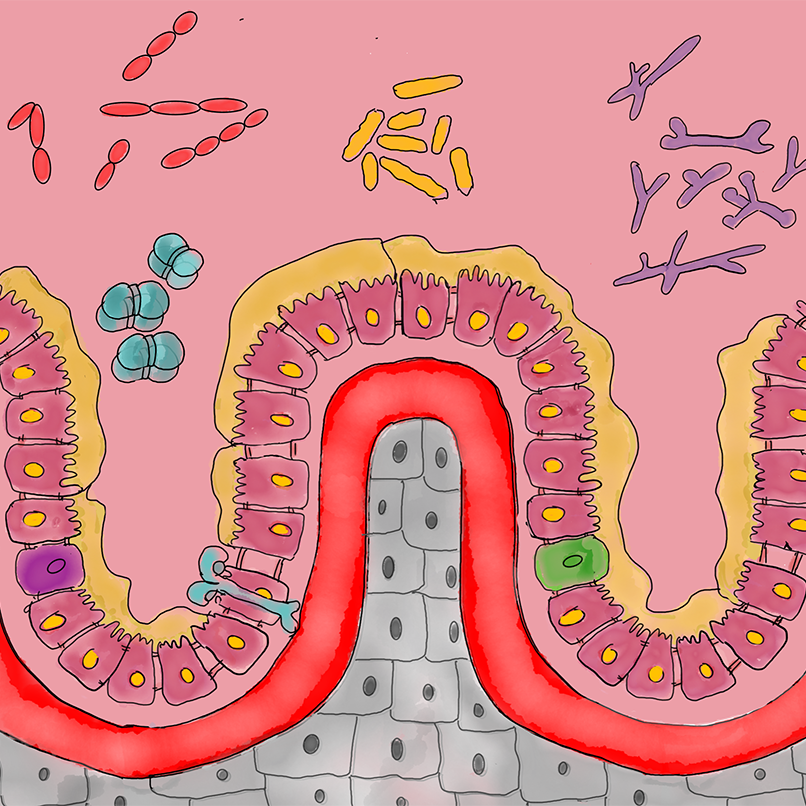
X. TARGET: Microbiome/Gastrointestinal system (n=6)
Microbiome/GIT concerns a category of agents specifically targeting the gastrointestinal system. E.g. think of a trial in which rifaximin – a broad-spectrum oral antibiotic – is being tested (MICRO-PD), one in which fecal microbiota transplantation (a technique in which intestinal microbiota are transferred from a healthy donor to the patient) and a trial that aims to treat anxiety in Parkinson’s Disease with a multistrain probiotic.

XI. MECHANISM OF ACTION: Neurotrophic factors (n=3)

XII. MECHANISM OF ACTION: Non-dopaminergic symptom relief (n=41)
Neurodegeneration in patients with Parkinson’s disease is not limited to the substantia nigra where dopamine is produced. Other areas of the brain are also affected to a greater or lesser extent. Non-dopaminergic symptom relief concerns trials investigating the impact on neurotransmitters, other than dopamine, including (nor)adrenergic, serotonergic, glutamatergic, and cholinergic systems. Which is an as broad category as it sounds 🙂 Apart from motor symptoms this category also addresses autonomic dysfunction, neuropsychiatric symptoms, and systemic symptoms such as pain and fatigue. Examples include a trial with clonidine, an adrenergic receptor agonist, to treat impulse control disorder, and a trial with droxidopa for fatigue. Droxidopa is a precursor to the neurotransmitter noradrenaline.

XIII. Other (n=16)

XIV. TARGET: aSN (n =5)
aSN is short for alpha-synuclein, a protein of 140-amino acids expressed abundantly in the brain. Five trials are using small molecules to target a-synuclein, e.g. mannitol and memantine. Mannitol supposedly is an aggregation inhibitor of aSN, which is taken to be good news since the aggregation of aSN supposedly contributes to pathogenesis*. Memantine is a partial NMDA (N-methyl-d-aspartate receptor) receptor antagonist which means that it fits in the receptor and partly deactivates it supposedly preventing alpha-synuclein accumulation at the same time. Abnormal expressions of both alpha-synuclein and N-methyl-d-aspartate (NMDA) receptors are observed in Parkinson’s.
* Whether through a loss-of-function (LOF) of the native protein and/or a toxic gain-of-function (GOF) and/or something else altogether is a current topic of debate that even made it to the book Brain Fables.
Hope visualised
I have played around with the ‘drug pipeline’ data and made some vizzes. Below each of the 145 trials is assigned to one of the 14 drug categories just described. The colours in the circles give additional information about the purpose of the drug. You can interact with the visualisation, e.g. by making a selection of the data you want to see (just not so easy on a mobile phone though).
DMT stands for disease-modifying therapies. These are drugs that are being tested in the hope they will be able to alter the course of Parkinson’s. The others are symptomatic treatments divided by the type of symptom which is addressed. Symptomatic treatments don’t change the course of the disease but do (temporarily) mask the ongoing neurodegenerative process by reducing the symptom burden.
Below you can find a few more vizzes. They were made with Tableau Public which is free for all to download and explore if you’d like to give it a try yourself (Or better even: try an open source data visualisation programme while you are at it).
Click image to visit the interactive version
In the viz above you can see that – in general – the amount of participants increases as the phase of the clinical trial moves upwards. Want to know more about the ins and outs of clinical trials? Then go visit the website ‘Clinical trials explained in plain English‘. I myself also greatly enjoyed reading the book ‘Clinical trials. What patients and healthy volunteers need to know‘.
Click image to visit the interactive version
For this viz, I added some columns to the data of Kevin et al. Whereas in the original data, all sponsors/collaborators are grouped together in one field, I have given each contributor it’s own field so I can visualise the data per sponsor/collaborator. Also, I have manually looked up the country of the headquarters of each sponsor/collaborator in order to make this geographic viz. In Kevin et al.’s article, there is also a geographic visualisation of the location where the trials are held (where the participants go to). These locations aren’t always equal to the location of the headquarters of a company.
Part 3. Hope, shared FAIRly
The Hope list that Kevin himself has compiled – bit.ly/ParkinsonsHopeList – is open to explore, to visualise, to enrich, to change. The data take the central stage.
In the wonderful publication by Kevin and co-authors, the data are added as a supplementary file in PDF format (McFarthing et al, 2020). A PDF doesn’t really have the ‘Come on in, you’re welcome’ doormat at its front door. To build upon the data you first have to transform it into .csv or Excel. The question is: Should science be protected against changes? Isn’t science in fact the business of incremental change (as long as we correctly version and annotate the data)?
The thing is: In a publication, authors answer questions that you and I don’t necessarily have. But what if you’d like to see another cross-section of the data? What if you’d like to improve the data quality by adding your own knowledge into a new column? What if you’d like to verify each data point? What if you’d like to make your own visualisation? What if you’d like to combine different datasets?
To increase the (reuse) potential of scientific research, research data are best shared in a way that makes them FAIR: Findable, Accessible, Interoperable, Reusable. At present, this isn’t mainstream yet and we often have to email the authors to retrieve the data. The ‘drug pipeline’ paper is just one of the many examples where the research data is made available, be it hidden as supplementary material in PDF format. But research data deserve a place and persistent identifier of their own. Some journals have data availability policies to speed up this type of research data sharing (e.g. PLOS ONE).
A change of paradigm
Until recently, the prevailing paradigm in science has been article-centric. For a long time, we simply couldn’t afford a data-centric approach. I myself have loads of paper lab notebooks in my attic that were never distributed because of the sheer physical volume they take up. Digital information, however, doesn’t take up physical space and is copied in an instant.
Time to reset science, right? Because in the end, it’s the research data that are the center of the scientific universe; not the articles. It’s the lists of raw hope – collected by (the likes of) Kevin – that are pure gold. All conclusions and discussions based upon any data should humbly circle around such data; not the other way around.
Anyone who – while reading this post – suddenly realises that he/she has accidentally been hiding research data, can start choosing a data repository – such as Zenodo – to upload their ‘datasets of hope’. In this way, your research data will get a citable DOI, become more visible/findable, easier to reuse, and you can see the downloads. Uploading new versions of major revisions without overwriting the old ones would allow for trends in time to be analysed.
Because ….
The best thing to do with your data will be thought of by someone else
— rufuspollock (@rufuspollock) April 20, 2017
Thank you @brianglanz for sharing#OpenData #OpenKnowledge pic.twitter.com/p16sNGXjpY
So now it’s your turn. How would like to build upon Kevin’s list of hope?
I’m overly curious to find out!
Sparks
Source
McFarthing, K., Buff, S., Rafaloff, G., Dominey, Th., Wyse, R.K., Stott, S.R.W. (2020). Parkinson’s Disease Drug Therapies in the Clinical Trial Pipeline: 2020’. Journal of Parkinson’s Disease, vol. 10, no. 3, pp. 757-774. https://doi.org/10.3233/JPD-202128
* This was part III of a series of blogs in which I put a Parkyvist in the spotlight. In my definition, a Parkyvist is someone who has a diagnosis of Parkinson’s disease and who noticeably uses his/her talents to end Parkinson’s. Other Parkyvists in this series:
PS
Oops, I notice that I will definitely need to work on the gender gap I have been creating here 😉 I have Sara Riggare, Omatola Thomas and Simone Rensen on my list. Moreover, I’d also like to bridge other gaps (geographical, ethnic, etc.) and be as inclusive as possible. Your tips are most welcome. Luckily, in the meantime, the parkyvists wordwide just keep on going.







